JAC Board Class 9th Science Solutions Chapter 2 Is Matter Around Us Pure
JAC Class 9th Science Is Matter Around Us Pure InText Questions and Answers
Page 15
Question 1.
What is meant by a pure substance?
Answer:
A pure substance consists of a single type of particles and it cannot be separated into other kinds of matter by any separation process.
Question 2.
List the points of difference between ho-mogeneous and heterogeneous mixtures.
Answer:
Differences between homogeneous and heterogeneous mixtures:
| Homogeneous mixture | Heterogeneous mixture |
| It has no visible boundary or boundaries of separation between its constituents. | It has visible boundary boundaries of separation between its constituents. |
| It has a uniform composition. | It does not have a uniform composition. |
| They form solutions. | They form suspensions colloids. |
| The particle size is very small. | The particle size is larger. |
| For example, sugar + water → sugar solution. | For example, sugar + sand. |
Page 18
Question 1.
Differentiate between homogeneous and heterogeneous mixtures with examples.
Answer:
See answer 2 above.
![]()
Question 2.
How are sol, solution and suspension different from each other?
Answer:
Comparison among properties of true solution, colloidal solution and suspension:
| Property | True solution | Colloidal solution | Suspension |
| Appearance | Hetero geneous and transparent. | Hetero geneous and translucent. | Hetero geneous and opaque. |
| Particle size | lnm (10-9 m) | lnm – 1000 nm | 1000 lnm (10-6m) |
| Visibility | Particles are not visible even with a powerful microscope. | Particles can be seen with a high power microscope. | Particles can be seen with naked eyes. |
| Stability | Stable | Stable | Unstable |
| Diffusion | Diffuse rapidly | Diffuse slowly | Do not diffuse |
| Filterability | Passes through filter paper, e.g., sodium chloride dissolved in water. | Passes through filter paper, e.g., blood. | Can be separated by filter paper, e.g., mud water. |
Question 3.
To make a saturated solution, 36g of sodium chloride is dissolved in lOOg of water at 293K. Find its concentration at this temperature.
Answer:
Mass of solute (sodium chloride) = 36g
Mass of solvent (water) = 100 g
Mass of solution = Mass of solute + Mass of solvent = 36g + 100g =136g
\(\frac{ Mass of solute}{Mass of solution}\) × 100 ⇒ \(\frac{36 \times 100}{136}\)
= 26.47%
Page 24
Question 1.
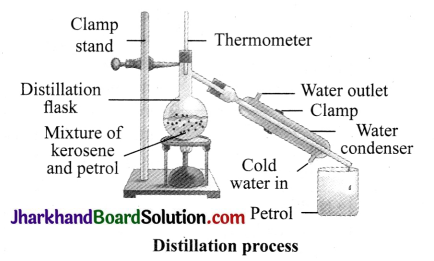
How will you separate a mixture containing kerosene and petrol (difference in their boiling points is more than 25°C), which are miscible with each other?
Answer:
Set up the apparatus as shown in the figure. Take the given mixture in a distillation flask. Heat the mixture slowly, keeping a close watch at the thermometer. At a certain point, temperature becomes constant. Petrol vaporises first as it has lower boiling point. It condenses in the condenser and is collected from the condenser outlet. Stop heating when the temperature further starts rising. Kerosene is left behind in the distillation flask.
Question 2.
Name the technique to separate:
(a) Butter from curd
(b) Salt from sea – water
(c) Camphor from salt
Answer:
(a) Butter from curd – Centrifugation
(b)Salt from sea – water – Evaporation
(c) Camphor from salt – Subiimation
Question 3.
What type of mixtures are separated by the technique of crystallisation?
Answer:
Crystallisation technique is used to purify a solid with some impurities in it. For example, purification of salt obtained from sea – water.
Page 25
Question 1.
Classify the following as chemical or physical changes: cutting of trees, melting of butter in a pan, rusting of almirah, boiling of water to form steam, passing of electric current, through water and the water breaking down into hydrogen and oxygen gases. making of fruit salad with raw fruits, dissolving common salt in water, burning of paper and wood
Answer:
- Physical change:
- Cutting of trees
- Melting of butter in a pan
- Boiling of water to form steam
- Dissolving common salt in water
- Making a fruit salad with raw fruits
- Chemical change:
- Rusting of almirah
- Passing of electric current through water and the water breaking down into hydrogen and oxygen gases
- Burning of paper and wood
Question 2.
Try segregating the things around you as pure substances or mixtures.
Answer:
- Pure substances: Water, sugar, gold, copper wire, salt, ice, etc.
- Mixtures: Steel, plastic, paper, talc, milk, air, ink, soda water, lemonade, bread, etc.
JAC Class 9th Science Is Matter Around Us Pure Textbook Questions and Answers
Question 1.
Which separation techniques will you apply for the separation of the following?
(a) Sodium chloride from its solution in water.
(b) Ammonium chloride from a mixture containing sodium chloride and ammonium chloride.
(c) Small pieces of metal in the engine oil of a car.
(d) Different pigments from an extract of flower petals.
(e) Butter from curd.
(f) Oil from water.
(g) Tea leaves from tea.
(h) Iron pins from sand.
(i) Wheat grains from husk.
(j) Fine mud particles suspended in water.
Answer:
(a) Crystallisation/Evaporation
(b) Sublimation
(c) Filtration
(d) Chromatography
(e) Centrifugation
(f) Separating funnel
(g) Filtration
(h) Magnetic separation
(i) Winnowing
(j) Decantation/Sedimentation
Question 2.
Write the steps you would use for making tea Use the words solution, solvent, solute, dissolve, soluble, insoluble, filtrate and residue.
Answer:
Take a cup of water in a kettle as solvent and heat it. When the solvent boils, add sugar in it which is the solute. Heat it till entire sugar dissolves. Water and sugar form a solution. Then, add some tea leaves in this solution. Boil the contents, add milk which is also soluble in this mixture, and boil again. Filter the tea with the help of a strainer. The tea collected in cup is the filtrate and the tea leaves collected in the strainer is residue.
![]()
Question 3.
Pragya tested the solubility of three different substances at different temperatures and collected the data as given below (results are given in the following table, as grams of substance dissolved in 100 grams of water to form a saturated solution).
| Temperature in K Solubility | |||||
| Substance Dissolved | 283 | 293 | 313 | 333 | 353 |
| Potassium nitrate | 21 | 32 | 62 | 106 | 167 |
| Sodium chloride | 36 | 36 | 36 | 37 | 37 |
| Potassium chloride | 35 | 35 | 40 | 46 | 54 |
| Ammonium chloride | 24 | 37 | 41 | 55 | 66 |
(a) What mass of potassium nitrate would be needed to produce a saturated solution of potassium nitrate in 50 grams of water at 313 K?
(b) Pragya makes a saturated solution of potassium chloride in water at 353K and leaves the solution to cool at room temperature. What would she observe as the solution cools? Explain.
(c) Find the solubility of each salt at 293K. Which salt has the highest solubility at this temperature?
(d) What is the effect of change of temperature on the solubility of a salt?
Answer:
(a) Mass of potassium nitrate (KNO3) needed to produce a saturated solution of KNO3 in 100 grams of
water at 313 K = 62 g
Mass of KNO3 needed in 50 g of
water at 313 K = \(\frac{62.0 \times 50}{100}\)
= 31.0g
(b) As solution cools, potassium chloride gets crystallised. This is because the solubility of a solid decreases with decrease in temperature.
(c) At 293K, solubility of KNO3 is 32, NaCl is 36, KCL is 35 andNH4Cl is 37. Ammonium chloride has the highest solubility at 293 K temperature.
(d) As the temperature increases, solubility increases and vice – versa.
Question 4.
Explain the following giving examples.
(a) saturated solution
(b) pure substance
(c) colloid
(d) suspension
Answer:
(a) Saturated solution: A solution in which no more solute can be dissolved at a given temperature is called a saturated solution.
(b) Pure substance: A pure substance consists of a single type of particles. It always has the same colour, taste or texture at a given temperature and pressure. For example, pure water is always colourless, odourless and tasteless.
(c) Colloid: A colloid is a solution in which the size of solute particles is bigger than those of a true solution. These particles cannot be seen with naked eyes as they are stable, e.g., ink, blood, smoke, milk, fog and cloud.
(d) Suspension: Suspension is a heterogeneous mixture. The particles of a suspension are greater than 1000 nm and are visible to naked eyes, e.g., chalk powder in water, paints, etc.
![]()
Question 5.
Classify each of the following as a homogeneous or a heterogeneous mixture: soda water, wood, air, soil, vinegar, filtered tea.
Answer:
- Homogeneous mixtures: Soda water, vinegar, filtered tea.
- Heterogeneous mixtures: Wood, soil, air.
Question 6.
How would you confirm that a colourless liquid given to you is pure water?
Answer:
By finding the boiling point of the given colourless liquid. If the given colourless liquid boils at exactly 373K at 1 atmosphere pressure, then it is pure water. This is because pure substances have fixed melting and boiling points.
Question 7.
Which of the following materials fall in the category of a ‘pure substance’?
(a) Ice
(b) Milk
(c) Iron
(d) Hydrochloric acid
(e) Calcium oxide
(f) Mercury
(g) Brick
(h) Wood
(i) Air
Answer:
Pure substances are: ice (a compound), iron (an element), hydrochloric acid (a compound), calcium oxide (a compound) and mercury (an element).
Question 8.
Identify the solutions among the following mixtures.
(a) Soil
(b) Sea water
(c) Air
(d) Coal
(e) Soda water
Answer:
Solutions are sea water, soda water and air.
Question 9.
Which of the following will show “Tyndall effect”?
(a) Salt solution
(b) Milk
(c) Copper sulphate solution
(d) Starch solution
Answer:
Milk and starch solution are colloids and will show Tyndall effect.
Question 10.
Classify the following into elements, compounds and mixtures.
Sodium, Soil, Sugar solution, Silver, Calcium carbonate, Tin, Silicon, Coal, Air, Soap, Methane, Carbon dioxide, Blood
Answer:
- Elements: Sodium, Silver, Tin, Silicon
- Compounds: Calcium carbonate, Meth – ane, Carbon dioxide
- Mixtures: Sugar solution, Soil, Coal, Air, Blood, Soap
Question 11.
Which of the following are chemical changes?
(a) Growth of a plant
(b) Rusting of iron
(c) Mixing of iron filings and sand
(d) Cooking of food
(e) Digestion of food
(f) Freezing of water
(g) Burning of a candle
Answer:
Chemical changes are growth of a plant, rusting of iron, cooking of food, digestion of food, burning of a candle.